Articles / New malaria vaccine: How much promise does it hold?


writer
Snow Medical Fellow & CSL Centenary Fellow, Burnet Institute

writer
Infectious Disease Researcher; Director and CEO, Burnet Institute; President, Australian Global Health Alliance, Pacific Friends of Global Health
Malaria, caused by infection with Plasmodium parasites and transmitted via mosquitoes, still causes many millions of cases and more than 600,000 deaths annually, the majority in children under five and pregnant women in Africa.
This is the second malaria vaccine to be endorsed by the WHO, following a vaccine called RTS,S/AS01 in 2021.
Both are much-needed tools in the global fight to eradicate malaria, with the potential to save millions of lives. But the new vaccine offers some unique advantages.
Both the newly endorsed vaccine, R21, and the earlier vaccine, RTS,S, aim to protect against malaria by blocking the Plasmodium falciparum parasite before it can cause infection. They do this by targeting the parasite during the bite from a mosquito.
Called “sub-unit” vaccines, they contain a single protein from P. falciparum, the circumsporozoite protein, which coats the surface of the parasite. The vaccines aim to drive an immune response that can subsequently recognise the circumsporozoite protein, and kill the parasite before it causes infection.
It’s a bit like how most COVID vaccines target the “spike” protein, a protein on the surface of SARS-CoV-2 (the virus that causes COVID) to block the virus.
Both malaria vaccines are administered with an adjuvant, a special formula to help activate the immune response. They contain the same parasite protein target, but are designed with different amounts of parasite protein, and unique adjuvants.
New results from phase 3 trials of R21 show the vaccine can prevent up to 75% of malaria cases over the year following vaccination. This is in areas where malaria occurs seasonally and when the vaccine is used in combination with other preventive measures including drugs. It’s important to note this data has not yet been peer-reviewed.
Peer-reviewed phase 3 trials of RTS,S showed the vaccine can prevent around 56% of cases in the year following vaccination across a large number of malaria infection sites.
These vaccines haven’t been tested directly against each other, so we can’t say conclusively whether one vaccine is more effective than the other, though it seems possible that R21 offers enhanced protection.
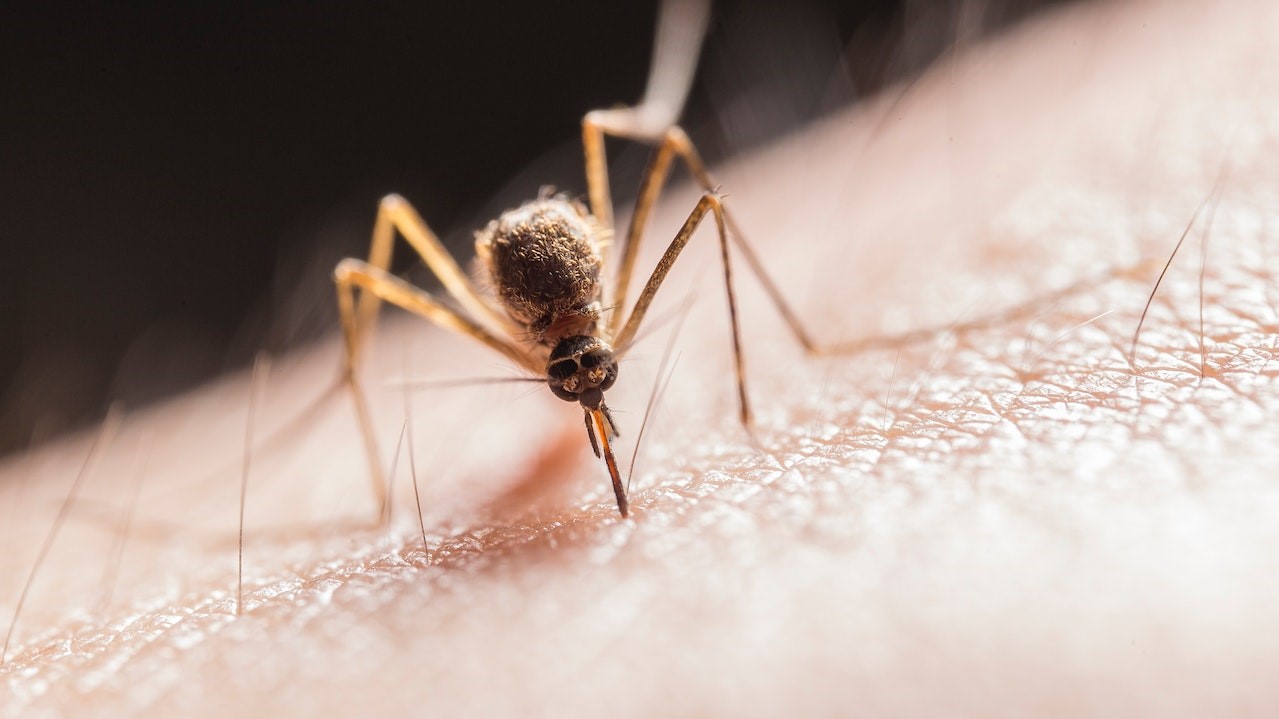
R21 also has significant advantages when it comes to manufacturing, supply and cost, with 100-200 million doses predicted to be available annually from existing manufacturers in India. Because of this, and with the ability for manufacturing to be expanded to other malaria-endemic countries, the cost of R21 will be less than US$5 a dose (compared to only around 15 million doses for RTS,S at nearly twice the cost per dose).
The availability of the R21 vaccine should make access and rollout across malaria-endemic areas feasible in the coming years.
The short answer is no, but it promises to help a great deal.
Despite the impressive protection reported from the R21 vaccine, challenges remain. The vaccine requires three doses over three months in the first instance, followed by a booster dose one year later. It also seems likely that yearly boosters will be required to maintain protection.
As we’ve seen in the rollout of COVID vaccines, boosters are challenging to administer even in areas such as Australia with very strong health systems. To date, neither of the malaria vaccines have been integrated within other childhood vaccination programs, placing an additional burden on communities.
Further, R21 is yet to be tested in an area of very high malaria risk. Results from the RTS,S vaccine flag this as a concern, because in areas of high malaria transmission there is a risk that previous infection can block the efficacy of vaccines, leaving those most at risk with reduced protection.
Finally, both endorsed vaccines only target one type of malaria, that caused by P. falciparum parasites. This is the most deadly form of malaria, so it’s understandable that vaccine efforts have focused on this species.
But it’s important to note that these vaccines don’t protect against other types of malaria, most especially Plasmodium vivax, which causes the most disease in southeast Asia.
Further research and development will be needed to develop second-generation malaria vaccines. Especially important will be vaccines that induce high levels of sustained protection without the need for yearly boosters, vaccines that protect against all malaria types (particularly P. vivax), and vaccines or additional drugs that can boost protection for children who have the highest malaria risk.
While not a silver bullet, the availability of two malaria vaccines is an exciting development.
We’ve seen with the COVID pandemic that vaccines can have a significant individual and public health impact when used alongside other tools. Likewise, it’s essential that malaria control strategies make integrated use of all available interventions.
The use of these two vaccines, with continued use of preventive drugs, insecticide-treated bed nets, fast and effective drug treatments, and other control tools, will make a big difference and save lives.
Many of us in the field wondered if we would ever see an approved malaria vaccine. As WHO director-general Tedros Adhanom Ghebreyesus said, “As a malaria researcher, I used to dream of the day when we would have a safe and effective vaccine against malaria. Now we have two.”![]()
Michelle Boyle, Snow Medical Fellow & CSL Centenary Fellow, Burnet Institute and Brendan Crabb, Director and CEO, Burnet Institute
This article is republished from The Conversation under a Creative Commons license. Read the original article.![]()

Menopausal Hormone Therapy - What Dose of Estrogen is Best?

Cardiovascular Benefits of GLP1s – New Evidence

Oral Contraceptive Pill in Teens

RSV and the Heart

writer
Snow Medical Fellow & CSL Centenary Fellow, Burnet Institute

writer
Infectious Disease Researcher; Director and CEO, Burnet Institute; President, Australian Global Health Alliance, Pacific Friends of Global Health

Modified but kept in place
Eliminated entirely without replacement
Maintained as is
Completely replaced with an alternative system
Listen to expert interviews.
Click to open in a new tab
Browse the latest articles from Healthed.
Once you confirm you’ve read this article you can complete a Patient Case Review to earn 0.5 hours CPD in the Reviewing Performance (RP) category.
Select ‘Confirm & learn‘ when you have read this article in its entirety and you will be taken to begin your Patient Case Review.
Menopause and MHT
Multiple sclerosis vs antibody disease
Using SGLT2 to reduce cardiovascular death in T2D
Peripheral arterial disease
